Proposed mechanism of action
5-isopropyloxycarbonyl-6-methyl-4-(2-nitrophenyl)-2-[(7-chloroquinolin-4-ylamino)butylamino] pyrimidine or 10r as it is referred to in the paper, is the candidate proposed as a treatment to Malaria caused by P. falciparum. The drug shows much promise by inhibiting DHFR-ts (dihydrofolate reductase-thymidylate synthase) which, is vital in DNA replication for P. falciparum.
The chemical 10r, has a similar proposed action to many commercially available malaria treatments. The mechanism of action blocks the DHFR-ts action through competitive inhibition binding and disrupting the production of other essential molecules including, dTMP used to make Thymidine nucleotides for DNA replication and the amino acid Glycine used in protein transcription(1).
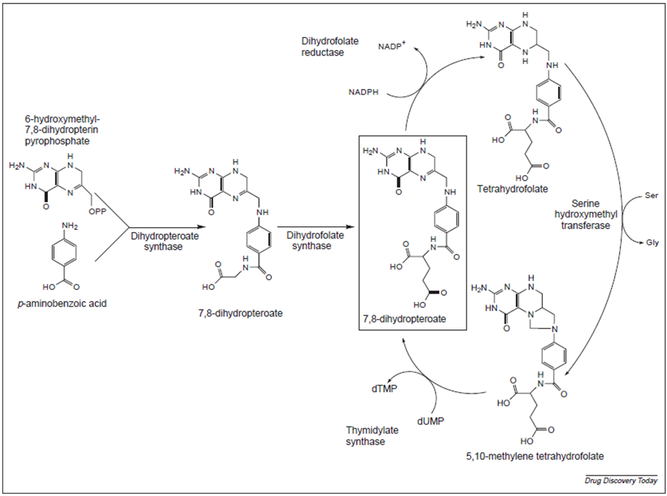
figure 3.2. the folate synthase pathway
The ribbon diagrams below demonstrate the similarities between human DHFR and P. falciparum DHFR-ts, The additional structure found on P. Falciparum DHFR-ts is a thymidylate synthase that allows the protein to produce dihydrofolate. The change in conformation caused by the additional subunit is significant enough that it allows DHFR-ts to be targeted specifically over human DHFR.
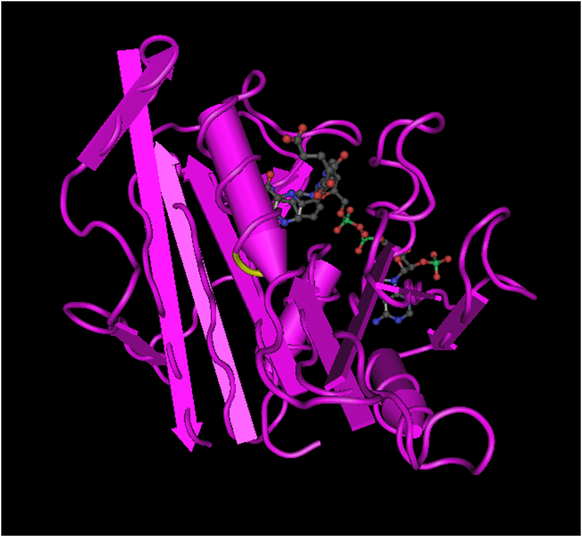
figure 3.3 Human DHFR
The ribbon diagrams above and below demonstrate the similarities between human DHFR and P. falciparum DHFR-ts, highlighted in the purple sections. The additional structure found on P. Falciparum DHFR-ts is a thymidylate synthase that allows the protein to produce dihydrofolate. The addintional subunit creates a conformational change and it is this creates a significant enough difference that it allows DHFR-ts to be targeted specifically over human DHFR.
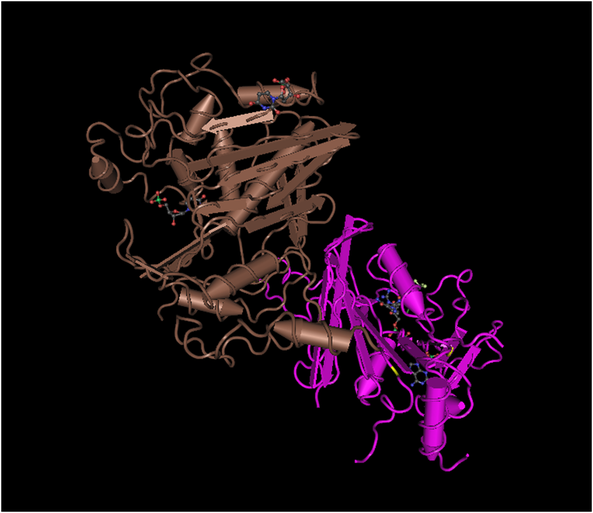
fig3.4
Plasmodium Falciparum DHFR-TS
Plasmodium Falciparum DHFR-TS
Both the human DHFR and P. falciparum DHFR subunit share many structural similarities. However, the protein found in P. falciparum contains a secondary subunit, Thymidylate Synthase (TS), in addition to the DHFR that from a multi-protein complex. The flanking TS and DHFR active sites in parasitic TS-DHFRs are distinct which makes it such a promising drug target, even though DHFR and TS are highly conserved in eukaryotes(2).
This is where problems arise as most common anti malaria drugs exhibit a low binding affinity to human DFHR. The new proposed drug has an extremely high specificity for P. falciparum DHFR-ts. Meaning a lower dose is required for the same effect and cytotoxic effects on the host are minimized(1).
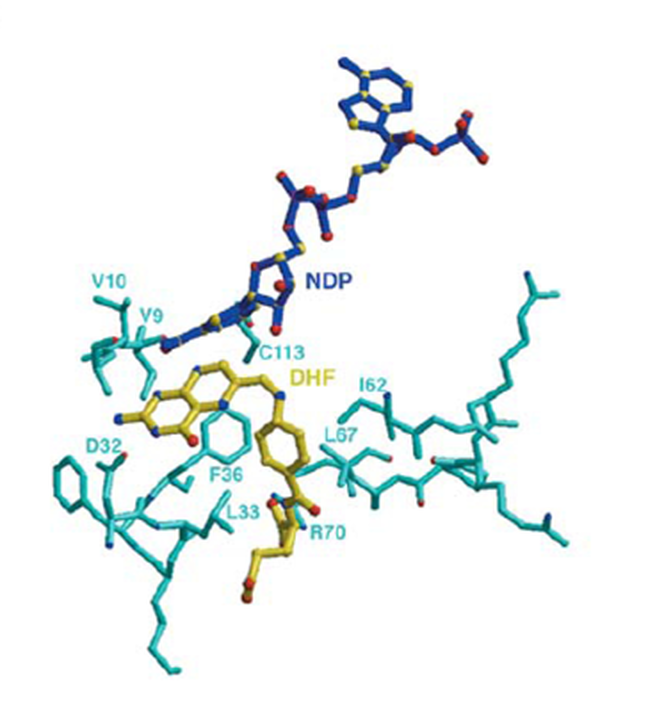
fig 3.5 DHFR-TS active residues with native DihydroFolate
Figure 3.5 highlights the interaction between the DHFR-TS and Dihydrofolate (DHF) its natural substrate and the active residues that interact to with the substrate to produce tetrahydrofolate
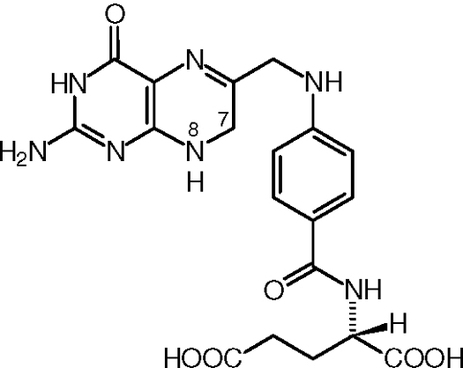
fig 3.6 Dihydrofolate
the compund below 10r is the proposed treatement, it shares many similar functional groups with DHF. 10r is a hybrid molecule of 7-chloroqunine and 5-isopropyloxycarbonyl-6-methyl-4-(2-nitrophenyl)-2-butylamino pyrimidine which share many simillar structural features with DHF. 5-isopropyloxycarbonyl-6-methyl-4-(2-nitrophenyl)-2-butylamino pyrimidine is thought to bind with DFHR-TS in a simillar function to DHF and whilst the 7-Chloroqunine with a highly electronegative Chlorine is thought to change the conformation of the DHFR-TS blocking its action.
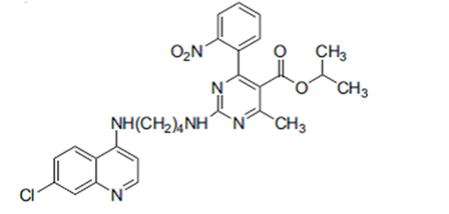
fig 3.7 10r
10r also has other bioogical acttivity that make it a promising candiadate, 10r aswell as some other compounds investigate where shown to inhibit the formation of B-hematin complex's. The B-hematin is a byproduct the plasmodium produce from feeding on blood vessels, the complex is formed to remove souble heme which, is toxic to to the malaria parasite(4).
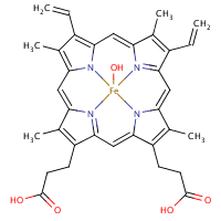
fig 3.8 hematin
1.Kamaljit Singh, Hardeep Kaur, Kelly Chibale, Jan Balzarini, Susan Little, Prasad V. Bharatam, 2-Aminopyrimidine based 4-aminoquinoline anti-plasmodial agents. Synthesis, biological activity, structure–activity relationship and mode of action studies, European Journal of Medicinal Chemistry, Volume 52, June 2012, Pages 82-97, ISSN 0223-5234, 10.1016/j.ejmech.2012.03.007.
3.7
2. Anderson, C.,(2005). Targeting DHFR in Parasitic Protozoa. Drug Discovery Today.10.121-128
3.2,3.5
3.F. Rodríguez, I. Rozas, M. Kaiser, R. Brun, B. Nguyen, W.D. Wilson, R.N. García, C. Dardonville, New Bis (2-aminoimidazoline) and bisguanidine DNA minor groove binders with potent in vivo antitrypanosomal and antiplasmodial activity, J. Med. Chem. 51 (2008) 909e923.
4. Cody, V. et al. (1992) Crystal structure determination at 2.3 Å of recombinant human dihydrofolate reductase ternary complex with NADPH and methotrexate-γ-tetrazole. Anticancer Drug Des. 7, 483–491
